How Many Covalent Bonds Can Each Carbon Atom Form
How Many Covalent Bonds Can Each Carbon Atom Form - Covalent bonds are bonds that are formed between nonmetals. Group 5a form 3 bonds; In a covalent bond, two molecules share a couple of electrons. Carbon has an unusual ability to bond to itself. Group 6a form 2 bonds; The atomic number of carbon is 6. Atoms of carbon can bond with each other or with atoms of other elements. With hydrogen, nitrogen, oxygen, and other heteroatoms. Form long c −c chains,. Web one carbon atom forms four covalent bonds with four hydrogen atoms by sharing a pair of electrons between itself and each hydrogen (h) atom.
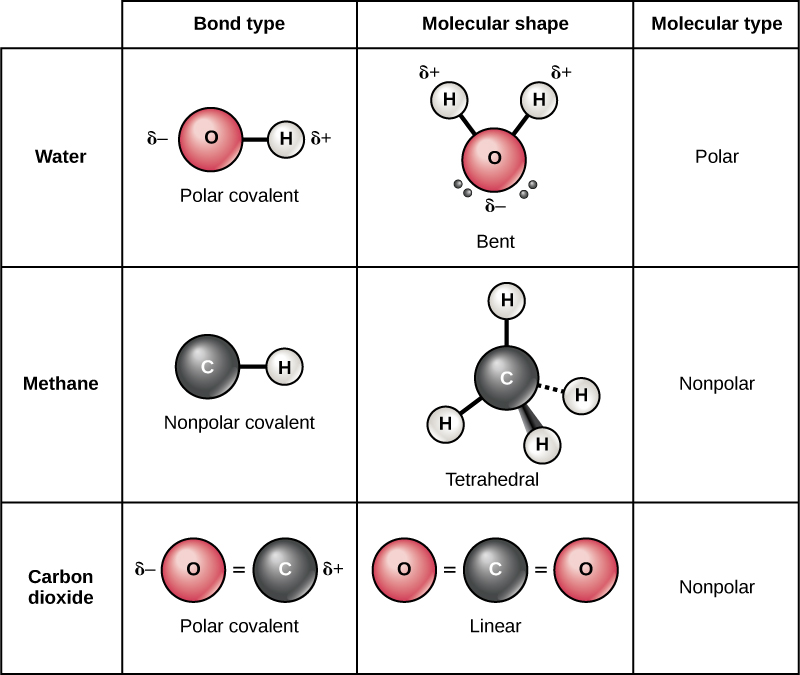
Web typically, the atoms of group 4a form 4 covalent bonds; For example, water, h 2 o, has two single bonds, one between each hydrogen atom and the oxygen atom (fig. Covalent bonds are bonds that are formed between nonmetals. With hydrogen, nitrogen, oxygen, and other heteroatoms. Web carbon can form four covalent bonds. Web 4 rows typically, the atoms of group 4a form 4 covalent bonds; Web carbon contains four electrons in its outer shell. Web a molecule can have multiple single bonds. A triple bond results when two atoms share three electron pairs to form three covalent. In a covalent bond, two molecules share a couple of electrons.
Carbon has an unusual ability to bond to itself. And group 7a form one bond. Web carbon can form four covalent bonds. Web a molecule can have multiple single bonds. Web carbon has four valence electrons, so it can achieve a full outer energy level by forming four covalent bonds. A bond composed of two electrons , one from each of the two. Web because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. Web 4 rows typically, the atoms of group 4a form 4 covalent bonds; Web well, carbon can form up to four covalent bonds. Web typically, the atoms of group 4a form 4 covalent bonds;
Bonding A Level Notes
Therefore, it can form four covalent bonds with other atoms or molecules. Group 5a form 3 bonds; Atoms of carbon can bond with each other or with atoms of other elements. Web carbon contains four electrons in its outer shell. Web typically, the atoms of group 4a form 4 covalent bonds;
Carbon — Role and Importance to Life Expii
In a covalent bond, two molecules share a couple of electrons. Group 5a form 3 bonds; Web because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. Web the carbon atom has unique properties that allow it to form covalent bonds to as many as four different atoms, making.
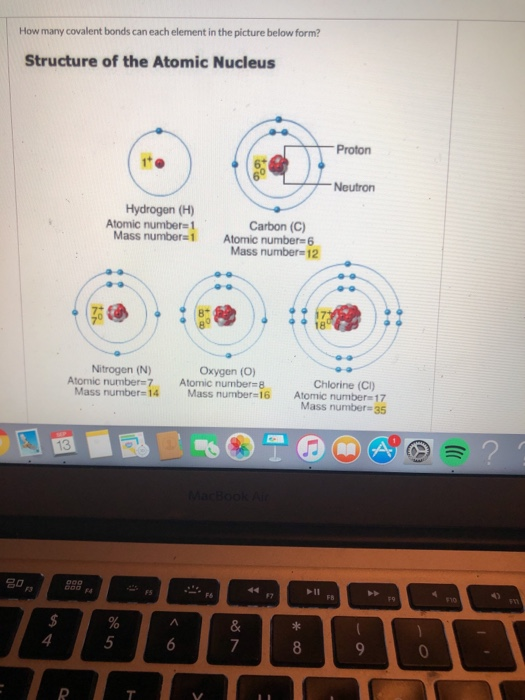
Solved How many covalent bonds can each element in the
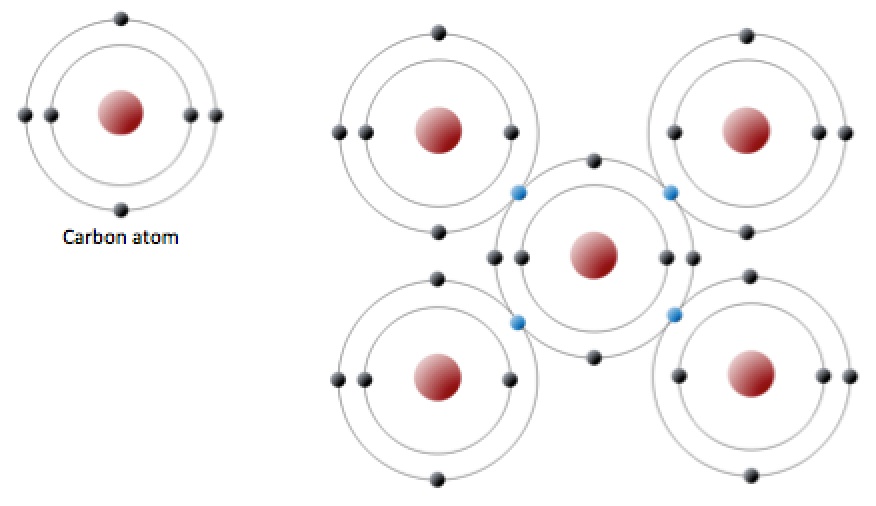
Carbon atoms can join together to make molecules. A triple bond results when two atoms share three electron pairs to form three covalent. Web a molecule can have multiple single bonds. Web carbon has four valence electrons, so it can achieve a full outer energy level by forming four covalent bonds. The number of electrons required to obtain.
We ‘Share’ the Oil field with others in Europe, having a claim to the
This means that it has six protons and six electrons. The number of electrons required to obtain. A triple bond results when two atoms share three electron pairs to form three covalent. Web each carbon atom forms four covalent bonds. Group 5a form 3 bonds;
2.2 Chemical Bonds Anatomy & Physiology
This enables carbon to share four. For example, water, h 2 o, has two single bonds, one between each hydrogen atom and the oxygen atom (fig. Carbon has an unusual ability to bond to itself. Web carbon has four valence electrons, so it can achieve a full outer energy level by forming four covalent bonds. Group 6a form 2 bonds;
Atoms, Isotopes, Ions, and Molecules The Building Blocks · Biology
Web each carbon atom forms four covalent bonds. Web two atoms of the same element can be joined together in covalent molecules. Web because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. Carbon has an unusual ability to bond to itself. A bond composed of two electrons ,.
How Many Single Bonds Can Carbon Form fredhughesdesign
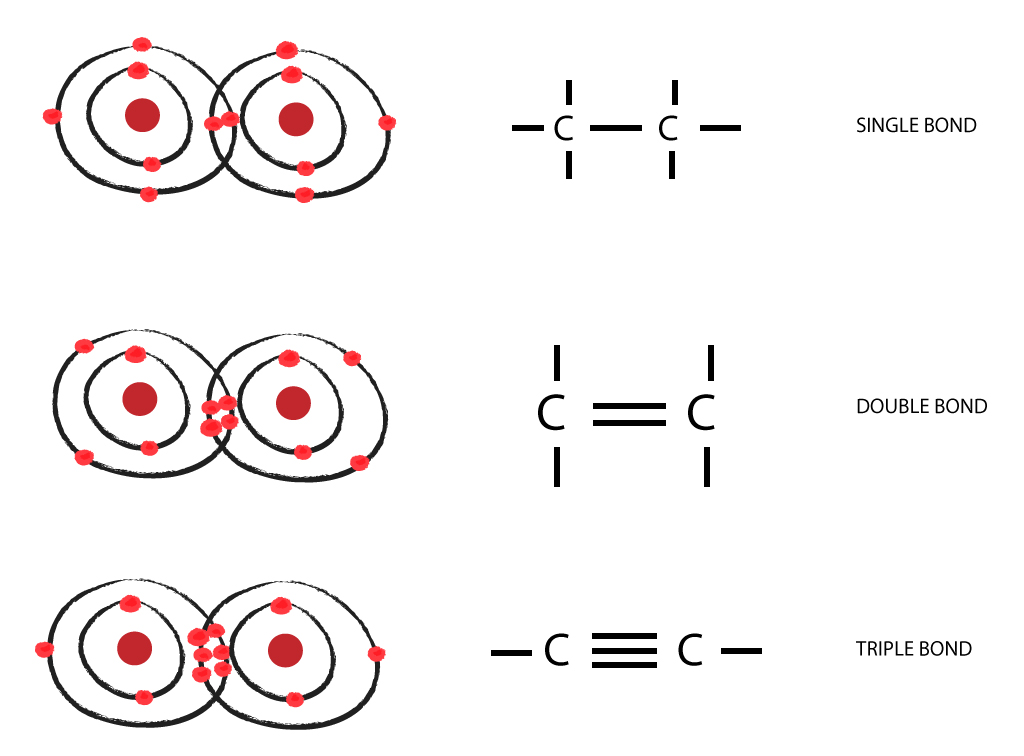
Web two atoms of the same element can be joined together in covalent molecules. Group 6a form 2 bonds; The simplest organic carbon molecule is methane (ch 4),. Web a double bond is formed when two atoms use two electron pairs to form two covalent bonds; For example, water, h 2 o, has two single bonds, one between each hydrogen.
2.2 Bonding and Lattices Physical Geology
Form long c −c chains,. With hydrogen, nitrogen, oxygen, and other heteroatoms. Web well, carbon can form up to four covalent bonds. This enables carbon to share four. Atoms of carbon can bond with each other or with atoms of other elements.
Covalent Bonding (Biology) — Definition & Role Expii
When it bonds only with hydrogen, it forms compounds called. In a covalent bond, two molecules share a couple of electrons. The most common form is the single bond : This means that it has six protons and six electrons. A triple bond results when two atoms share three electron pairs to form three covalent.
Carbon to Carbon Single, Double & Triple Bonds Surfguppy
This enables carbon to share four. And group 7a form one bond. For example, water, h 2 o, has two single bonds, one between each hydrogen atom and the oxygen atom (fig. When it bonds only with hydrogen, it forms compounds called. Web carbon can form four covalent bonds.
Web A Double Bond Is Formed When Two Atoms Use Two Electron Pairs To Form Two Covalent Bonds;
Carbon atoms can join together to make molecules. For example, water, h 2 o, has two single bonds, one between each hydrogen atom and the oxygen atom (fig. This enables carbon to share four. The number of electrons required to obtain.
When It Bonds Only With Hydrogen, It Forms Compounds Called.
Web a molecule can have multiple single bonds. The atomic number of carbon is 6. Web carbon has four valence electrons, so it can achieve a full outer energy level by forming four covalent bonds. Carbon has an unusual ability to bond to itself.
Form Long C −C Chains,.
Group 6a form 2 bonds; Web each carbon atom forms four covalent bonds. The most common form is the single bond : And group 7a form one bond.
In A Covalent Bond, Two Molecules Share A Couple Of Electrons.
Atoms of carbon can bond with each other or with atoms of other elements. The bonds may be single, double, or triple. A bond composed of two electrons , one from each of the two. Web because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules.


.PNG)