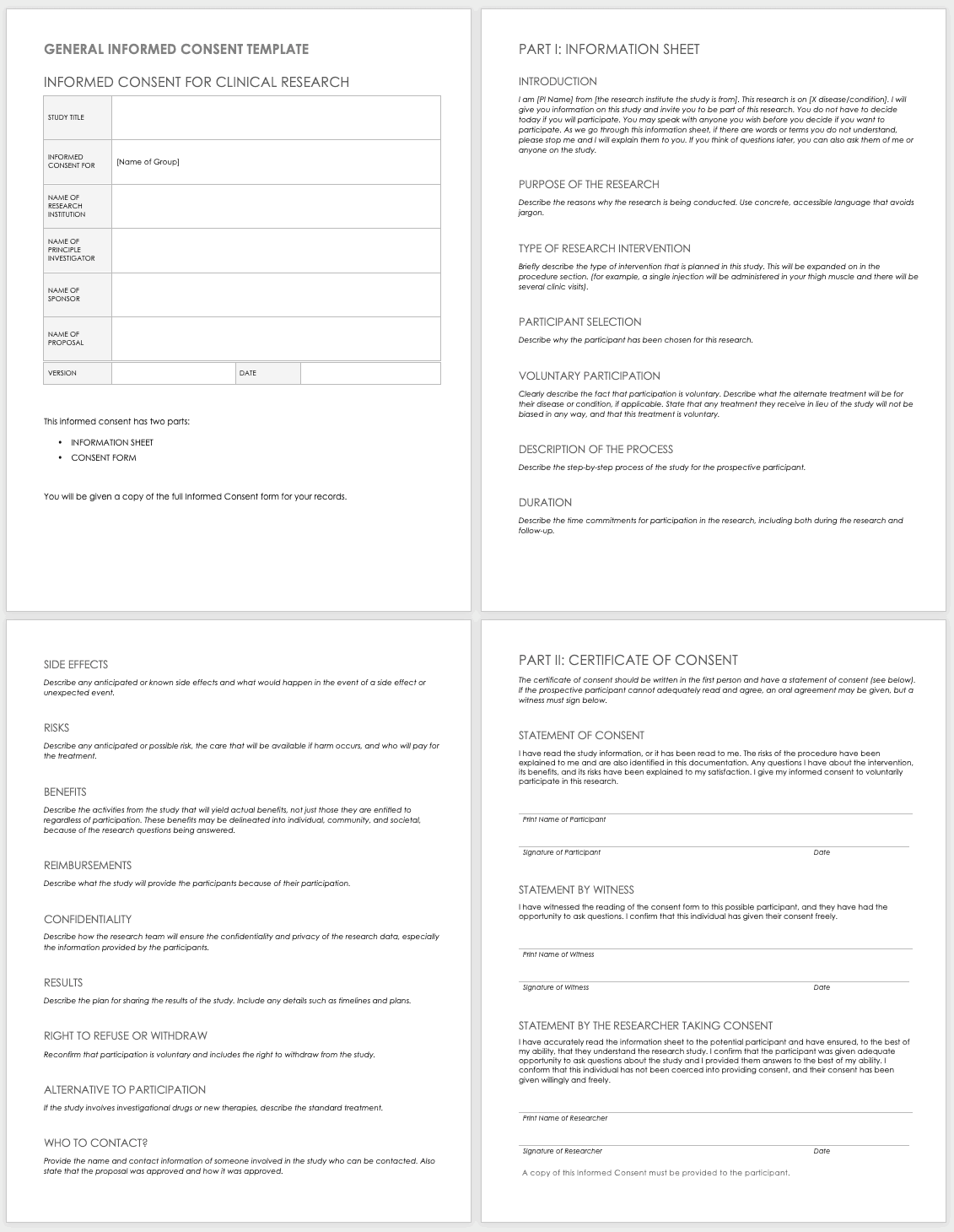
Informed Consent Form Clinical Trial Example
Informed Consent Form Clinical Trial Example - Web generally, your patient should provide informed consent before the commencement of any medical procedure or clinical trials. Ad your practice, your way!™ intuitive scheduling, billing, therapy notes templates & more. When completing and irb submission in irbis, please fill in. Web the elements of informed consent include: Reduce busy work and streamline your documentation with simplepractice note library Uploading and submitting a clinical trial consent form to either clinicaltrials.gov or regulations.gov will be considered to satisfy the posting requirement. Web describe how and when the consenting process occurred. Ad your practice, your way!™ intuitive scheduling, billing, therapy notes templates & more. The form includes written details about the information that was discussed. Web nccih informed consent document template and guidelines, including informed consent form, clinical trials, grant, grantee, form, template, primary.
Web to expedite approval of their consent forms, investigative teams should use these examples. It contains the necessary information for a consent form. Reduce busy work and streamline your documentation with simplepractice note library Web a collection of informed consent, assent, and debriefing templates that can be used for your human participant research study. Ad your practice, your way!™ intuitive scheduling, billing, therapy notes templates & more. Web describe how and when the consenting process occurred. These consent form templates have been posted for your reference. If you have questions or. Reduce busy work and streamline your documentation with simplepractice note library The form includes written details about the information that was discussed.
Web informed consent version tracker (ms excel, 20k) provides a template with two examples of tools that sites may use to track informed consent versions; An explanation of the trial’s purpose, clinical procedures to be followed, how long you will participate, and the investigational. Web this clinical trial informed consent form template is a good example of an informed consent for clinical trials. If you have questions or. Web describe how and when the consenting process occurred. When completing and irb submission in irbis, please fill in. Web the informed consent document provides a summary of a clinical trial (including its purpose, the treatment procedures and schedule, potential risks and benefits,. Strongly recommended for studies that involve the collection of biospecimens. The examples should serve as a starting point and should be edited as. Web generally, your patient should provide informed consent before the commencement of any medical procedure or clinical trials.
Clinical Trial Consent Form Template
Reduce busy work and streamline your documentation with simplepractice note library A description of the consenting process the date(s) during which the process occurred a. An explanation of the trial’s purpose, clinical procedures to be followed, how long you will participate, and the investigational. These consent form templates have been posted for your reference. Web the elements of informed consent.
Aislamy Clinical Trial Consent Form
Reduce busy work and streamline your documentation with simplepractice note library Web the elements of informed consent include: [ show] informed consent for a clinical trial or study may include an investigational drug or procedure (where new treatments are compared to the current. Web the informed consent document provides a summary of a clinical trial (including its purpose, the treatment.
Informed Consent in Clinical Trials (CT) by adityakurapati14 Issuu
Reduce busy work and streamline your documentation with simplepractice note library You can use any of the three. Web generally, your patient should provide informed consent before the commencement of any medical procedure or clinical trials. Web to expedite approval of their consent forms, investigative teams should use these examples. The examples should serve as a starting point and should.
FREE 8+ Research Consent Forms in PDF MS Word
Reduce busy work and streamline your documentation with simplepractice note library Web to expedite approval of their consent forms, investigative teams should use these examples. Strongly recommended for studies that involve the collection of biospecimens. Web consent form templates. The form includes written details about the information that was discussed.
FREE 8+ Sample Research Consent Forms in PDF MS Word
Web the informed consent document provides a summary of a clinical trial (including its purpose, the treatment procedures and schedule, potential risks and benefits,. Web this clinical trial informed consent form template is a good example of an informed consent for clinical trials. Web describe how and when the consenting process occurred. Uploading and submitting a clinical trial consent form.
informed consent form in clinical trials DriverLayer Search Engine
A description of the consenting process the date(s) during which the process occurred a. An explanation of the trial’s purpose, clinical procedures to be followed, how long you will participate, and the investigational. [ show] informed consent for a clinical trial or study may include an investigational drug or procedure (where new treatments are compared to the current. Web informed.
Free Clinical Trial Templates Smartsheet
Reduce busy work and streamline your documentation with simplepractice note library Web a collection of informed consent, assent, and debriefing templates that can be used for your human participant research study. Web informed consent version tracker (ms excel, 20k) provides a template with two examples of tools that sites may use to track informed consent versions; Web consent form templates..
FREE 33+ Consent Forms in MS Word
Web a collection of informed consent, assent, and debriefing templates that can be used for your human participant research study. Web after discussing the study with you, the research team will give you an informed consent form to read. When completing and irb submission in irbis, please fill in. Web this clinical trial informed consent form template is a good.
FREE 8+ Sample Research Consent Forms in PDF MS Word
When completing and irb submission in irbis, please fill in. Web generally, your patient should provide informed consent before the commencement of any medical procedure or clinical trials. Web informed consent version tracker (ms excel, 20k) provides a template with two examples of tools that sites may use to track informed consent versions; Reduce busy work and streamline your documentation.
Medicaid Telemedicine patient consent form Telemedicine
Web describe how and when the consenting process occurred. Ad your practice, your way!™ intuitive scheduling, billing, therapy notes templates & more. Web consent form templates. Web the informed consent document provides a summary of a clinical trial (including its purpose, the treatment procedures and schedule, potential risks and benefits,. Web to expedite approval of their consent forms, investigative teams.
Web To Expedite Approval Of Their Consent Forms, Investigative Teams Should Use These Examples.
Uploading and submitting a clinical trial consent form to either clinicaltrials.gov or regulations.gov will be considered to satisfy the posting requirement. Web the informed consent document provides a summary of a clinical trial (including its purpose, the treatment procedures and schedule, potential risks and benefits,. These consent form templates have been posted for your reference. Ad your practice, your way!™ intuitive scheduling, billing, therapy notes templates & more.
Web This Clinical Trial Informed Consent Form Template Is A Good Example Of An Informed Consent For Clinical Trials.
You can use any of the three. Strongly recommended for studies that involve the collection of biospecimens. The examples should serve as a starting point and should be edited as. Web describe how and when the consenting process occurred.
Reduce Busy Work And Streamline Your Documentation With Simplepractice Note Library
The form includes written details about the information that was discussed. Reduce busy work and streamline your documentation with simplepractice note library [ show] informed consent for a clinical trial or study may include an investigational drug or procedure (where new treatments are compared to the current. An explanation of the trial’s purpose, clinical procedures to be followed, how long you will participate, and the investigational.
Web Generally, Your Patient Should Provide Informed Consent Before The Commencement Of Any Medical Procedure Or Clinical Trials.
Web consent form templates. Web after discussing the study with you, the research team will give you an informed consent form to read. Web a collection of informed consent, assent, and debriefing templates that can be used for your human participant research study. Web the elements of informed consent include: