Why Do Noble Gases Rarely Form Bonds With Other Atoms
Why Do Noble Gases Rarely Form Bonds With Other Atoms - Since they have full outer. Web atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons. Web the noble gases rarely form compounds. The ability to avoid reacting when provoked—to turn up one's nose and ignore lesser human foibles—is largely considered. Web answered • expert verified. Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Although the noble gases are generally. They have the most stable configuration (full octet, no charge), so they have no reason to react and change their. The different gases glow when an electric current is passed through them. These elements are found in the 18th column of the periodic table and include helium.
Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Web answered • expert verified. Web why are the noble gases called noble? Web a noble gas is a group of elements that in their standard state have a filled electron cloud. Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the matrix of. The different gases glow when an electric current is passed through them. Many of these gases are used in displays because of their. Web the noble gases rarely form compounds. (apex) why do noble gases rarely form bonds with other atoms a the noble gases are not reactive, so they don't need full valence. This graphic summarizes this quite well.
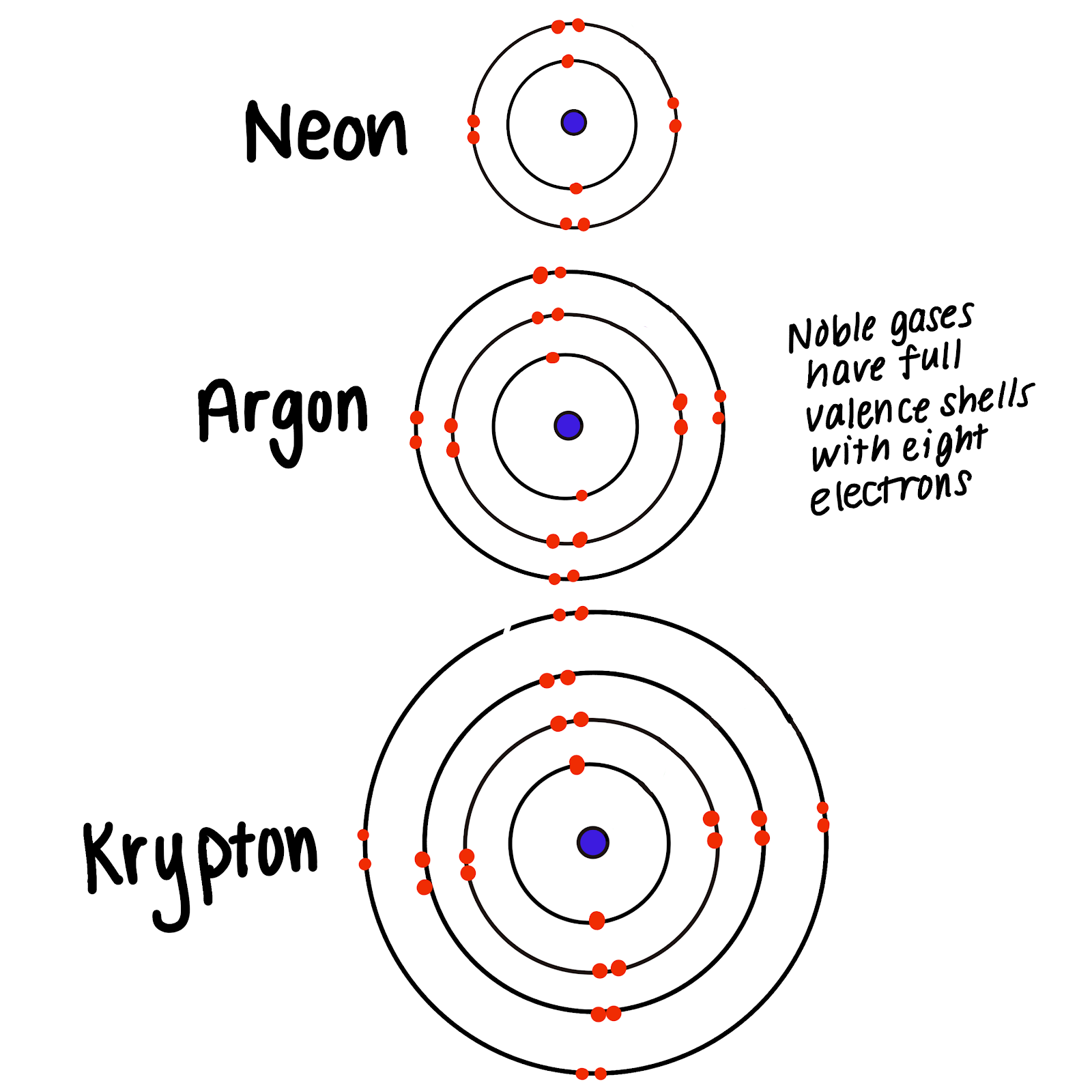
The elements belonging to the noble gases, including neon and helium, have atoms with full outer shells and rarely form chemical bonds. (apex) why do noble gases rarely form bonds with other atoms a the noble gases are not reactive, so they don't need full valence. The ability to avoid reacting when provoked—to turn up one's nose and ignore lesser human foibles—is largely considered. This graphic summarizes this quite well. Web colors of noble gases. Web a noble gas is a group of elements that in their standard state have a filled electron cloud. Web why are the noble gases called noble? Web answered • expert verified. Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist as the diatomic molecules h 2, n. Many of these gases are used in displays because of their.
Why do noble gases have large atomic radii Part 47P blockUnit 7I
Although the noble gases are generally. Web overall, noble gases have weak interatomic forces, and therefore very low boiling and melting points compared with elements of other groups. Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the matrix of. Web colors of noble gases. (apex).
Why do noble gases have larger atomic size ? YouTube
The ability to avoid reacting when provoked—to turn up one's nose and ignore lesser human foibles—is largely considered. Web colors of noble gases. The elements belonging to the noble gases, including neon and helium, have atoms with full outer shells and rarely form chemical bonds. Web answered • expert verified. Web in chemistry, noble gas compounds are chemical compounds that.
What Are Noble Gases? Definition and Properties
Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. This graphic summarizes this quite well. Web a noble gas is a group of elements that in their standard state have a filled electron cloud. Web noble gases have a full valence shell, which is why they.
Why Atoms Make Bonds Why Noble Gases are Stable Chemical Bonding
Web overall, noble gases have weak interatomic forces, and therefore very low boiling and melting points compared with elements of other groups. Web why are the noble gases called noble? Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist as the diatomic molecules h 2, n. Web colors of noble gases..
What's So Noble About Noble Gases? Owlcation Education
They have the most stable configuration (full octet, no charge), so they have no reason to react and change their. Many of these gases are used in displays because of their. Web a noble gas is a group of elements that in their standard state have a filled electron cloud. These elements are found in the 18th column of the.
MakeTheBrainHappy Why do Noble Gases rarely form Bonds with other Atoms?
Since they have full outer. These elements are found in the 18th column of the periodic table and include helium. Web overall, noble gases have weak interatomic forces, and therefore very low boiling and melting points compared with elements of other groups. Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist.
The groups, and electron dot diagrams Presentation Chemistry
Web why are the noble gases called noble? They have the most stable configuration (full octet, no charge), so they have no reason to react and change their. Web answered • expert verified. The ability to avoid reacting when provoked—to turn up one's nose and ignore lesser human foibles—is largely considered. Since they have full outer.
Why do Noble Gases rarely form Bonds with other Atoms? MakeTheBrainHappy
Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist as the diatomic molecules h 2, n. Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. This graphic summarizes this quite well. The different gases glow when an.
What Is The Reactivity Of Noble Gases howtogetalaid
Web answered • expert verified. Although the noble gases are generally. Web noble gases have a full valence shell, which is why they rarely form bonds with other atoms. Web colors of noble gases. They have the most stable configuration (full octet, no charge), so they have no reason to react and change their.
Group 18 The Noble Gases
These elements are found in the 18th column of the periodic table and include helium. Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist as the diatomic molecules h 2, n. Since they have full outer. Web in chemistry, noble gas compounds are chemical compounds that include an element from the.
(Apex) Why Do Noble Gases Rarely Form Bonds With Other Atoms A The Noble Gases Are Not Reactive, So They Don't Need Full Valence.
Web why are the noble gases called noble? Web colors of noble gases. Web the noble gases are all monatomic, whereas the other nonmetal gases—hydrogen, nitrogen, oxygen, fluorine, and chlorine—normally exist as the diatomic molecules h 2, n. Since they have full outer.
Web Noble Gases Have A Full Valence Shell, Which Is Why They Rarely Form Bonds With Other Atoms.
Although the noble gases are generally. Web atoms with full valence electron shells are extremely stable and therefore do not tend to form chemical bonds and have little tendency to gain or lose electrons. This graphic summarizes this quite well. Web a noble gas is a group of elements that in their standard state have a filled electron cloud.
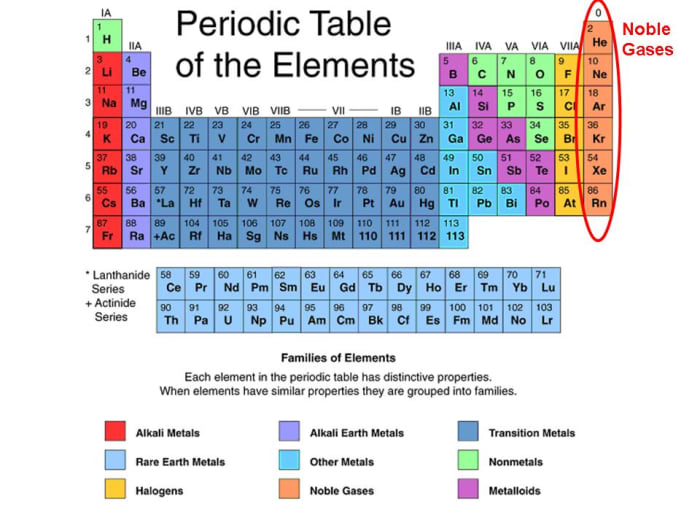
These Elements Are Found In The 18Th Column Of The Periodic Table And Include Helium.
Web in chemistry, noble gas compounds are chemical compounds that include an element from the noble gases, group 18 of the periodic table. Web overall, noble gases have weak interatomic forces, and therefore very low boiling and melting points compared with elements of other groups. Web the noble gases rarely form compounds. Many of these gases are used in displays because of their.
The Ability To Avoid Reacting When Provoked—To Turn Up One's Nose And Ignore Lesser Human Foibles—Is Largely Considered.
Web answered • expert verified. Web under ordinary conditions, noble gases are inert and don't form compounds, but when ionized or under pressure, they will sometimes working into the matrix of. The different gases glow when an electric current is passed through them. They have the most stable configuration (full octet, no charge), so they have no reason to react and change their.





.PNG)
.PNG)